CE Marking for Product Modifications and Variants: Compliance, Re-Assessment & Change Management Guide
If you manufacture or export products to Europe, you already know this truth: CE marking is not a one-time event. It’s a lifecycle responsibility. This is especially true for manufacturers managing CE compliance for exporters to Europe across evolving product lines.
Yet this is where many companies get into trouble.
They get CE marking for the original product. Then marketing asks for a new version. Engineering changes a component. A customer wants a customized variant. A supplier changes a material. A factory shifts production.
And suddenly the question appears:
“Do we need to redo CE marking for this change?”
Sometimes the answer is no. Sometimes it is absolutely yes. And sometimes the risk of getting it wrong is not just commercial—it’s legal.
This guide explains how CE marking for product modifications and CE marking for product variants really works, how to decide whether changes affect compliance, how to perform a CE change impact assessment, what documentation must be updated, what mistakes companies make, and how exporters use Qcert360 to manage CE compliance for design changes safely and efficiently.
What CE Marking Really Means for Manufacturers (Beyond the Sticker)
CE marking means you, as the manufacturer or legal responsible party, declare that your product complies with all applicable EU directives or regulations and continues to comply throughout its lifecycle. It is not a certificate you “get once.” It is a continuous legal obligation tied to the product design and configuration.
In business terms, CE marking:
• Is a legal declaration, not a marketing badge
• Covers design, materials, components, and manufacturing
• Must remain valid for every version placed on the EU market
• Transfers legal responsibility to the manufacturer or importer
• Can be challenged by authorities at any time
Once you understand this, CE compliance management for changing products looks very different.
Why Product Modifications and Variants Are a CE Compliance Risk
Every product change can potentially affect safety, performance, or regulatory compliance—even if it seems small from an engineering or commercial perspective. Regulators and market surveillance authorities don’t care why the change was made. They care whether the product still complies.
Typical triggers:
• Component substitutions due to supply chain issues
• Software or firmware updates
• Mechanical design changes
• New sizes, models, or power ratings
• Material changes
• Production process changes
• Relocation of manufacturing
If you don’t manage this systematically, you lose control of CE marking for modified products.
What Counts as a Product Modification Under CE Marking?
A product modification is any change that could affect compliance with applicable EU directives or harmonized standards—even if the product name and function stay the same. The risk is not the change itself, but its impact on safety or conformity.
Common examples:
• Changing a power supply or motor
• Changing enclosure material or thickness
• Modifying control software
• Altering dimensions, load ratings, or temperature limits
• Changing critical suppliers or components
Some changes are harmless. Some are legally critical—especially in CE marking for machinery changes, EMC products, and electrical equipment.
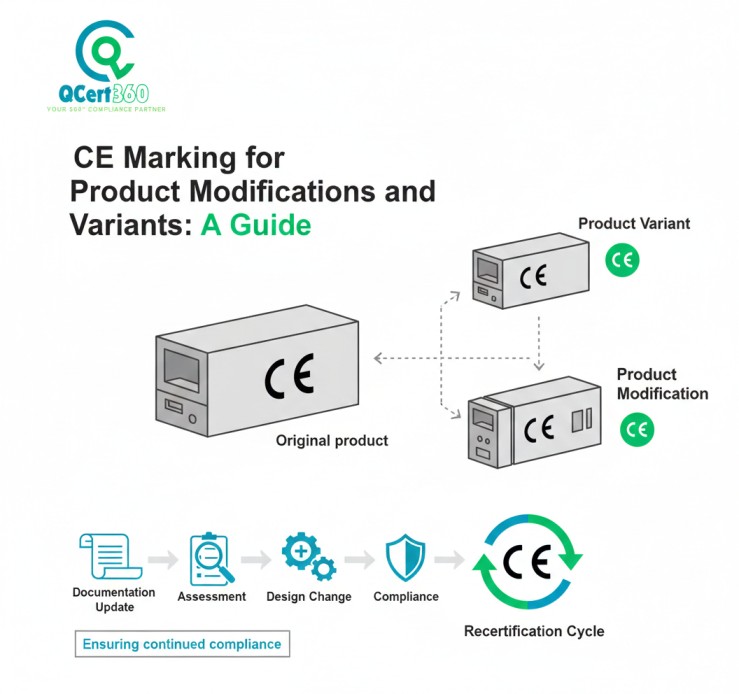
What Is a Product Variant in CE Marking and CE Compliance Terms?
A product variant is a different version of the same base product that shares a design platform but differs in certain characteristics such as size, power, configuration, or features. From a CE perspective, each variant must be demonstrably compliant.
Examples:
• Same machine in different sizes
• Same device with different voltage or power ratings
• Same product with optional modules
• Same product with different housings or accessories
Variants are not automatically covered just because the base model was compliant. This is why CE marking for product families requires a defined conformity strategy.
When Does a Product Change Require CE Re-Assessment or Re-Certification?
A change requires CE re-assessment when it can affect compliance with essential requirements, risk assessment, or harmonized standard conformity. The decision is not based on cost, convenience, or time pressure. It is based on safety and regulatory impact.
In simple terms:
• If the change can affect risk, performance, or safety, you must review CE compliance.
• If it cannot, you must still document why you believe it does not.
No documentation = no defense.
CE Marking Legal Responsibility After Product Changes: Who Is Accountable?
The legal responsibility for CE compliance always sits with the manufacturer or the entity placing the product on the EU market under their name. This does not change when you modify a product.
That means:
• You cannot blame the supplier
• You cannot blame the customer
• You cannot blame the factory
• You cannot blame the designer
If your name is on the Declaration of Conformity, you own the risk.
How to Perform a CE Change Impact Assessment (In Practice)
Every product modification or variant should go through a structured CE change impact assessment before release. This is not bureaucracy—it’s legal risk management.
A practical process includes:
• Describing the change clearly
• Identifying affected directives and standards
• Checking which essential requirements may be impacted
• Reviewing the risk assessment
• Reviewing test results and calculations
• Deciding whether re-testing or re-evaluation is needed
• Documenting the decision
This is your legal safety net.
Which EU Directives Are Most Sensitive to Product Modifications?
Some CE directives are especially sensitive to design or component changes because they are directly safety-critical.
Examples:
• Machinery Directive / Machinery Regulation
• Low Voltage Directive (LVD)
• EMC Directive (often triggers EMC re-testing after modification)
• Radio Equipment Directive (RED)
• Medical Devices Regulation (MDR)
• ATEX
In these frameworks, small changes can have big regulatory consequences.
The Role of Risk Assessment in CE Marking for Product Changes
The risk assessment is the heart of CE marking. Any change that can affect hazards, exposure, or risk reduction measures must trigger a risk review.
In practice:
• You update the risk assessment
• You check whether existing controls are still adequate
• You identify new hazards introduced by the change
• You decide whether additional measures or tests are required
If the risk assessment changes, the CE compliance basis changes.
How to Update CE Technical Documentation After Product Modifications
CE marking is not just about the product. It’s about the technical file that proves compliance. Any relevant change must be reflected in the documentation.
This may include:
• Updated drawings and BOMs
• Updated risk assessment
• Updated test reports
• Updated calculations
• Updated instructions and warnings
• Updated Declaration of Conformity
If your file doesn’t match the product, you must update CE technical documentation or you are not compliant.
When Do You Need New Testing or CE Re-Certification After a Change?
You need new testing or formal re-evaluation whenever a product change affects characteristics that were previously verified by testing or assessment. The decision must be based on documented impact analysis, not assumptions.
Typical examples include:
• EMC tests when electronics, PCB layout, clock frequencies, or shielding change
• Safety tests when materials, insulation, creepage/clearances, enclosure, or mechanical structure change
• RF tests when wireless modules, antennas, or RF paths change
• Performance tests when ratings, capacities, or declared performance parameters change
In some cases, partial or delta testing is enough. In others, full re-testing or re-certification is unavoidable. The key is to prove the decision with technical justification, not guesswork.
Common CE Marking Mistakes When Modifying Products or Creating Variants
Most CE compliance failures after product changes are not technical. They are organizational and procedural.
Common mistakes include:
• Letting engineering or purchasing change components without CE review which breaks the legal compliance chain
• Assuming “minor change = no impact” without analysis instead of doing a formal impact assessment
• Not updating the technical file so documentation no longer matches the product on the market
• Not updating the Declaration of Conformity even though the legal basis or product scope has changed
• Not controlling product variants and slowly drifting outside the tested and justified design space
• Treating CE marking as a one-time project instead of a continuous legal obligation
These mistakes don’t show up in the lab first. They show up when authorities or customers start asking questions.
CE Marking Case Study: When a “Small Change” Became a Big Compliance Problem
A machinery manufacturer had valid CE marking for a successful machine that was being sold across multiple EU countries. Due to supply chain disruptions, they replaced a motor and control unit with a different model, assuming it was a “like-for-like” change.
What Went Wrong
The change was made without a formal CE impact assessment:
- No structured conformity impact analysis was performed before the change
- EMC behavior changed due to different drive and control electronics
- Safety functions responded differently, affecting stopping time and fault reactions
- The technical file and risk assessment were not updated
- A machine that had been compliant on paper was no longer compliant in reality
What Happened
- A customer complaint triggered a market surveillance inspection by authorities
- Inspectors found that the machine no longer matched the original CE conformity basis
- The authorities identified 5 major non-compliances related to EMC and safety
- Sales were temporarily blocked in one EU country and distributors were instructed to stop deliveries
- The company faced the risk of fines, forced recalls, and wider EU restrictions
How Qcert360 Fixed It
With a focused regulatory recovery approach, Qcert360:
- Re-performed the full risk assessment for the modified machine configuration
- Identified the affected EU directives and harmonized standards (Machinery, EMC, and related standards)
- Coordinated targeted EMC and functional safety testing with an accredited laboratory
- Updated the technical file, test reports, and Declaration of Conformity
- Implemented a formal engineering change control process to prevent similar issues in the future
- Prepared the company for authority follow-up review within 6 weeks
The Result
- Sales resumed within 8 weeks after corrective actions and re-validation
- No fines or recalls were issued by the authorities
- The machine regained full CE compliance status
- Future product changes are now systematically assessed before release
- CE compliance became controlled and proactive instead of reactive and risky
How to Build a CE Change Management System That Actually Works
Smart manufacturers don’t treat CE change decisions as ad-hoc engineering debates. They build a formal CE compliance management system that makes decisions predictable, defensible, and auditable.
A good CE change management system includes:
• Mandatory CE review for any design, component, or supplier change so nothing slips through by accident
• A standard impact assessment checklist to quickly decide whether safety, EMC, RF, or regulatory compliance is affected
• Clear responsibility for CE decisions so it’s obvious who approves, who assesses, and who signs off
• Documented decision and approval workflow to create legal evidence that changes were properly evaluated
• Integration with engineering and purchasing change processes so CE is checked before changes are released, not after
When this is in place, CE compliance becomes routine, not risky, and product updates stop turning into compliance emergencies.
How to Manage CE Marking for Product Families and Variants Efficiently
You don’t always need a full CE project for every single variant, but you do need a structured CE marking strategy for product families. Without this, small changes quickly turn into big compliance risks.
Common, accepted approaches include:
• Define a “worst-case” or “representative” model for testing that covers the highest-risk configuration
• Use design rules and constraints for variants so future models stay within the tested and assessed boundaries
• Use technical justification to cover product families instead of re-testing everything unnecessarily
• Clearly document the conformity logic so authorities and auditors can understand how variants are covered
When this is done properly, you get faster time to market, lower testing cost, and stronger legal defensibility.
What Market Surveillance Authorities Actually Check in the CE Marking Process
Market surveillance authorities don’t start by asking for test reports. They start by checking whether the product in the market actually matches the documentation you claim supports it.
In practice, they typically check:
• Product identification and configuration to confirm the model, version, and components match what’s declared
• Technical file consistency to see whether drawings, descriptions, and reports describe the real product being sold
• Risk assessment to verify that real hazards were identified and properly controlled
• Change history to see what has been modified since first CE marking and wh ether impacts were assessed
• Traceability between product and documentation to prove that test reports, certificates, and files really belong to that product
If the product and the paperwork don’t match, the discussion usually ends there, regardless of how many test reports exist.
CE Marking Consulting for Product Changes and Variants by Qcert360
Qcert360 works with manufacturers and exporters to build CE compliance systems that survive product evolution, not systems that break every time a component or model changes.
Our CE Mark Support service includes:
• CE change impact assessment frameworks to decide quickly and correctly whether a change affects compliance
• Variant and product family conformity strategies to reduce unnecessary testing while staying legally safe
• Technical file structuring and updating so documentation always matches the product on the market
• Directive and harmonized standard analysis to ensure the right rules are applied to each product and variant
• Test strategy and lab coordination to avoid over-testing while still protecting compliance
• Ongoing CE compliance management so changes, suppliers, and regulations don’t slowly create hidden risks
The result is controlled change, lower compliance risk, and CE marking that stays valid as your products evolve.
Not Sure Whether Your Latest Product Change Affects CE Compliance?
Many companies only realize this when a customer or authority asks uncomfortable questions.
👉 Request a CE Change Impact Review from Qcert360
You’ll get a clear, documented answer on whether your modification or variant affects CE compliance.
Want to Make CE Compliance Part of Your Product Lifecycle (Not a Bottleneck)?
If you regularly introduce new variants, versions, or upgrades, CE must become part of your design and change process.
👉 Book a CE Compliance Strategy Call with Qcert360
Get a practical approach to managing CE marking across your entire product portfolio.
CE Marking for Product Modifications and Variants – FAQs
- Do all product changes require CE re-certification?
No. But every change must go through a formal impact assessment. You must document whether the change affects safety, EMC, RF, or regulatory compliance and keep this in your technical file. - Can small component changes affect CE compliance?
Yes. Even small changes like a power supply, PCB component, or enclosure material can affect EMC, electrical safety, RF performance, or chemical compliance. - Do we need a new Declaration of Conformity for every variant?
Often yes. At minimum, the Declaration of Conformity must be updated to clearly cover the new variant or extended product family. - Can one test report cover multiple variants?
Sometimes, but only if technical equivalence can be clearly justified and documented in the technical file. Otherwise, additional testing is required. - Who decides whether a change affects CE compliance?
The manufacturer or the legally responsible entity always carries this responsibility, not the lab or the supplier. - What if a supplier changes a component without telling us?
You are still legally responsible for the product placed on the market. This is why supplier change control and incoming inspection are critical. - How long must we keep the technical file updated?
In most cases, at least 10 years after the last product is placed on the EU market, and it must be available to authorities on request. - Can Qcert360 review our existing CE files?
Yes. We perform technical file gap analysis, documentation checks, and compliance risk exposure reviews. - Is CE marking a one-time certification?
No. CE marking is a continuous legal obligation. Every product change, supplier change, or regulation update can affect compliance. - What’s the safest way to manage frequent product updates?
By implementing a formal CE change management and impact assessment process that reviews every change before release.
Our Services
ISO Standards
- ISO 9001 Certification
- ISO 14001 Certification
- ISO 45001 Certification
- ISO 22000 Certification
- ISO 17025 Certification
- ISO 27001 Certification
- ISO 13485 Certification
- ISO 20000-1 Certification
- ISO 41001 Certification
- ISO 22716 Certification
- ISO 50001 Certification
- ISO 22301 Certification
- ISO 29993 Certification
Product Certifications
Other international standards
- FSSC 22000 Certification
- HIPAA
- HACCP Certification
- SA 8000 Certification
- GMP Certification
- GDPR
- GDP Certification
- GLP Certification
- Certificate of Conformity
QCert360 provides a wide range of services including ISO certification, audit support, compliance consulting, and training. They specialize in helping businesses achieve global standards and certifications like ISO 9001, ISO 27001, ISO 14001, and many others. Their team ensures a seamless experience from consultation to certification, supporting clients at every stage.
The time it takes to achieve certification can vary depending on the complexity of the standard and the readiness of your organization. On average, it takes about 3 to 6 months. QCert360 works closely with clients to streamline the process, ensuring that all requirements are met efficiently and within a reasonable timeline.
QCert360 is a trusted partner with years of experience in helping businesses obtain international certifications. Their expert consultants provide tailored solutions, ensuring your organization not only meets but exceeds industry standards. With a customer-centric approach, they focus on offering end-to-end support to simplify the certification journey.
QCert360 serves a wide range of industries including manufacturing, healthcare, information technology, education, and services, among others. They customize their certification solutions to meet the unique requirements of each industry, ensuring relevance and compliance with global standards.
Yes, QCert360 provides ongoing support even after certification. They offer services like surveillance audits, recertification guidance, and consultancy to help maintain and improve your certification status. Their team ensures that your organization stays compliant and up-to-date with any changes in certification standards.
Getting started with QCert360 is simple. You can contact them via their website to request a consultation. Their team will assess your needs, discuss the best certification options for your business, and outline the steps involved. From there, they’ll guide you through the entire process, ensuring you’re prepared for certification.
QCert360 stands out due to its customer-focused approach, industry expertise, and comprehensive service offerings. Their team doesn’t just help you obtain certification but works to ensure your organization thrives in compliance with international standards. They also offer personalized consultation, making the process smoother and more efficient, ensuring long-term success for your business.
The cost of certification varies depending on factors such as the type of certification, the size and complexity of your organization, and the specific industry requirements. QCert360 offers competitive pricing and provides tailored quotes based on your unique needs. They ensure transparency and work with you to find the most cost-effective solution for your certification goals.
Yes, QCert360 offers internal audit services to help assess and improve your organization’s processes. Their expert auditors conduct thorough reviews of your systems and operations to ensure they meet required standards. They also provide actionable recommendations to help enhance efficiency and compliance, making sure you’re fully prepared for external audits.
If your organization doesn’t pass an audit or certification assessment, QCert360 works with you to understand the reasons for non-compliance and provides support to rectify the issues. They offer guidance on corrective actions and help you prepare for a re-assessment. Their goal is to ensure your organization meets the necessary standards for certification, and they will be by your side to make the process as smooth as possible.


